Due to a recent change of our website, the process for submitting refill requests online has now changed.
Please click on “Sign Up Today!†to create a new account, and be sure to download our NEW Mobile app!
Thank you for your patience during this transition
Get Healthy!
- Posted April 9, 2026
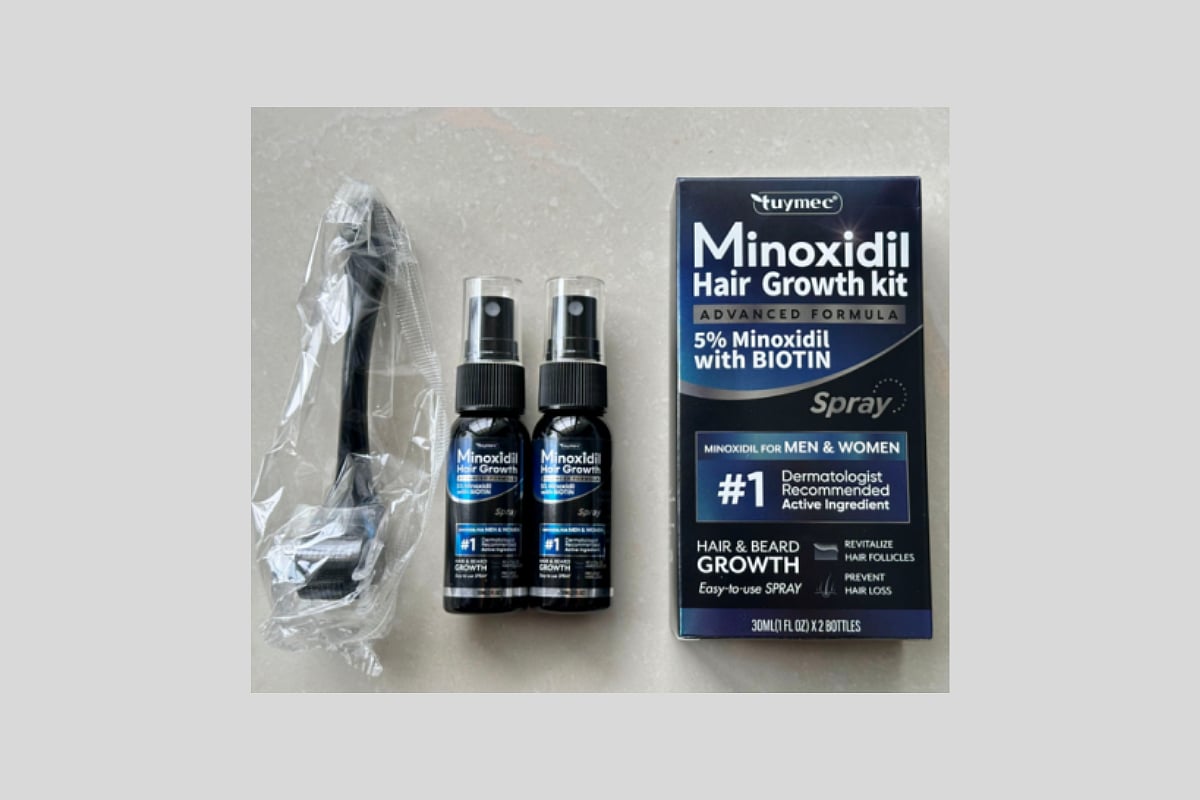
Hair Growth Product Recalled Over Child Poisoning Risk
A hair growth product is being recalled because it could pose a serious risk to kids.
The recall affects Tuymec Minoxidil Hair Growth Kits, which contain a 5% minoxidil spray with biotin, the U.S. Consumer Product Safety Commission (CPSC) said.
CPSC said the bottles are not child-resistant, which could increase the risk of injury or death if a child swallows the liquid inside.
Minoxidil is a medication used to treat hair loss, but it can be harmful if ingested.
As required by the Poison Prevention Packaging Act, products like this must be sold in child-resistant packaging.
The current packaging does not meet those safety standards, CPSC said.
The recall includes about 6,200 units sold on Amazon between June and August of last year for about $24.
The kits include two black spray bottles labeled with the following:
“Tuymec Minoxidil Hair Growth”
“Advanced Formula”
“5% Minoxidil with BIOTIN”
They were made in China and sold by DrHealBeauty of Japan, the CPSC said.
Anyone with the product should stow it out of children’s reach right away.
Contact DrHealBeauty to request a free replacement bottle with a child-resistant cap and then transfer the liquid into the new container.
Customers can reach out via email at drhealbeauty@gmail.com, or contact the company online at drhealbeauty.com and click “Recall Details” at the top of the page for more information.
No injuries have been reported.
More information
The Consumer Healthcare Products Association has more on child-resistant packaging.
SOURCE: U.S. Consumer Product Safety Commission, news recall, April 6, 2026
